 
Saving Earth Britannica Presents Earth’s To-Do List for the 21st Century.Britannica Beyond We’ve created a new place where questions are at the center of learning.100 Women Britannica celebrates the centennial of the Nineteenth Amendment, highlighting suffragists and history-making politicians.COVID-19 Portal While this global health crisis continues to evolve, it can be useful to look to past pandemics to better understand how to respond today.Student Portal Britannica is the ultimate student resource for key school subjects like history, government, literature, and more. Correlations between the viscosity B-coefficient of the Jones-Dole equation for aqueous solutions of alkali metal salts and the enthalpy of hydration of the.Britannica Explains In these videos, Britannica explains a variety of topics and answers frequently asked questions.Demystified Videos In Demystified, Britannica has all the answers to your burning questions.This Time in History In these videos, find out what happened this month (or any month!) in history.The greater the lattice enthalpy, the stronger the forces. #WTFact Videos In #WTFact Britannica shares some of the most bizarre facts we can find. Lattice enthalpy is a measure of the strength of the forces between the ions in an ionic solid.Britannica Classics Check out these retro videos from Encyclopedia Britannica’s archives.MgO the higher charge on Mg leads to a larger lattice energyĦ.It is a measure of the cohesive forces that bind ionic solids. Li 2O the higher charge on O 2– leads to a larger energy additionally, Cl – is larger than O 2– this leads to a larger interionic distance in LiCl and a lower lattice energy The lattice energy is the energy change upon formation of one mole of a crystalline ionic compound from its constituent ions, which are assumed to initially be in the gaseous state.MgO the higher charges on Mg and O, given the similar radii of the ions, leads to a larger lattice energy. 
MgO selenium has larger radius than oxygen and, therefore, a larger interionic distance and thus, a larger smaller lattice energy than MgO.The lattice energy formula can be stated as follows: LE kQ1Q2 / r Where, LE lattice energy k 2. The compounds with the larger lattice energy are Although lattice energy can be fairly complex, Coulomb’s law is frequently used to simplify it. Since the lattice energy is negative in the Born-Haber cycle, this would lead to a more exothermic reaction.Ĥ. 4008 kJ/mol both ions in MgO have twice the charge of the ions in LiF the bond length is very similar and both have the same structure a quadrupling of the energy is expected based on the equation for lattice energyĥ. The smaller the radius of the anion, the shorter the interionic distance and the greater the lattice energy would be. 
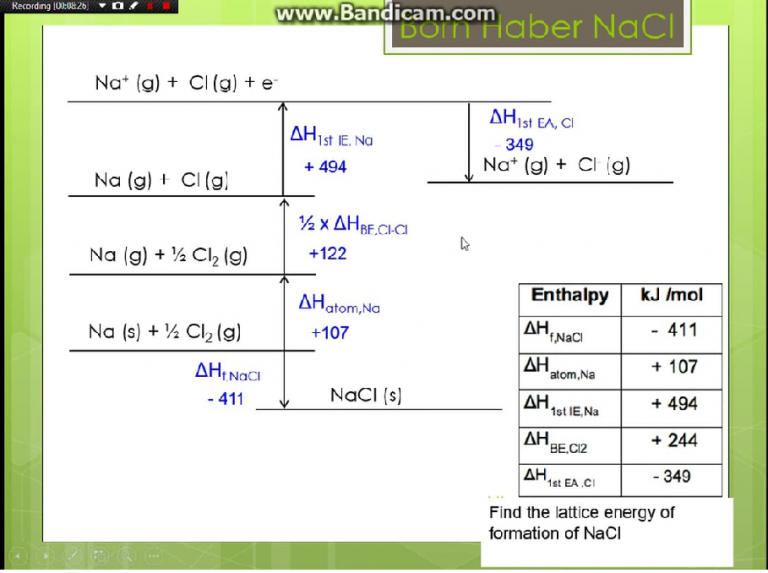
In the Born-Haber cycle, the more negative the electron affinity, the more exothermic the overall reaction.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
